Disrupting Modern Healthcare
The Psychedelics as Medicine Report: Third Edition
The Psychedelics as Medicine Report: Third Edition provides unparalleled consumer insights and market intelligence from experts and industry leaders, helping investors cut through the noise and identify real opportunities.
The Psychedelics as Medicine Report: Third Edition examines
- Proprietary insights from consumers and healthcare providers
- Deep dives into the most transformative compounds and companies
- Key regulations, transformative trends and commercial opportunities
Table of Contents
- Introduction
- Definition and Scope
- Executive Summary
- Key Trends
- Market Value
- Legislation and Regulation
- Consumer Attitudes
- Healthcare Providers’ Attitudes
- Spotlight on Health
- Psychedelics - Timeline of Key Developments
- Countries to Watch
- Psychedelic Profiles
- Key Psychedelics of Note
- Psychedelics As Medicines - Potential Therapies
- Psychedelics and Technology
- Psychedelics per Country
- Psychedelics and the Law
- Patents and Intellectual Property
- Therapists and Doctors
- Psychedelic Research
- Glossary
Key Trends
- Research is at a tipping point
- Psychedelics poised to treat many disorders
- Therapy will be the name of the game
- Legislation and regulation moving in the right direction
- Patients and doctors are ready for change
Market Value
- Approval landscape
- Projected growth of medical psychedelics market
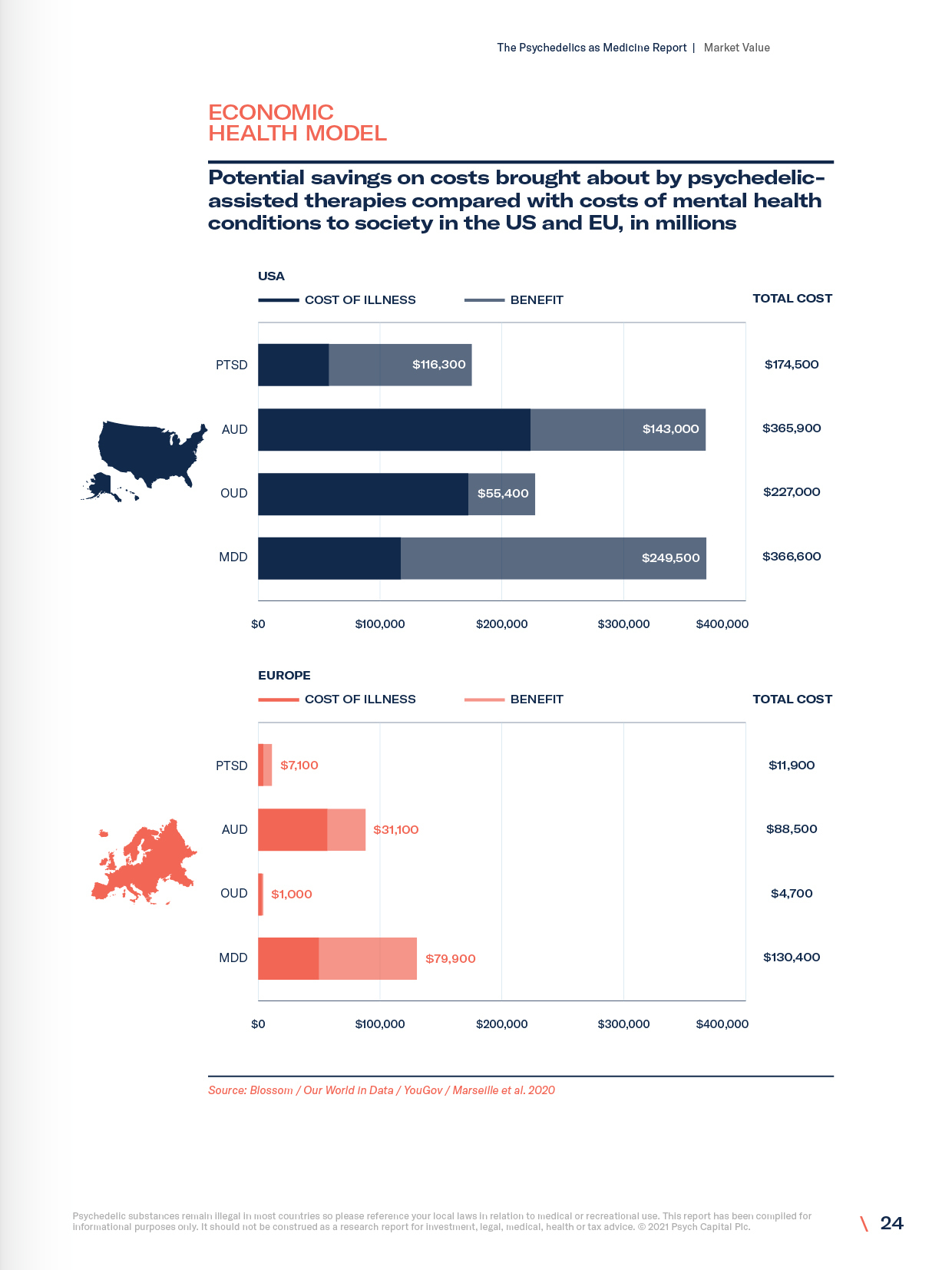
- The cost of illness
- Economic health model
- Mental health during the COVID-19 pandemic
Consumer Attitudes
- Consumer’s take on psychedelics as medicine
- The education opportunity
- Psychedelics of choice
- Influence of prior drug use on openness to psychedelic-assisted therapies
Healthcare Provider Attitudes
- State of mental health: Healthcare workers
- Psychedelic expertise of healthcare workers
- Healthcare workers' familiarity with psychedelic-assisted therapy
- Therapy: The benefits and challenges
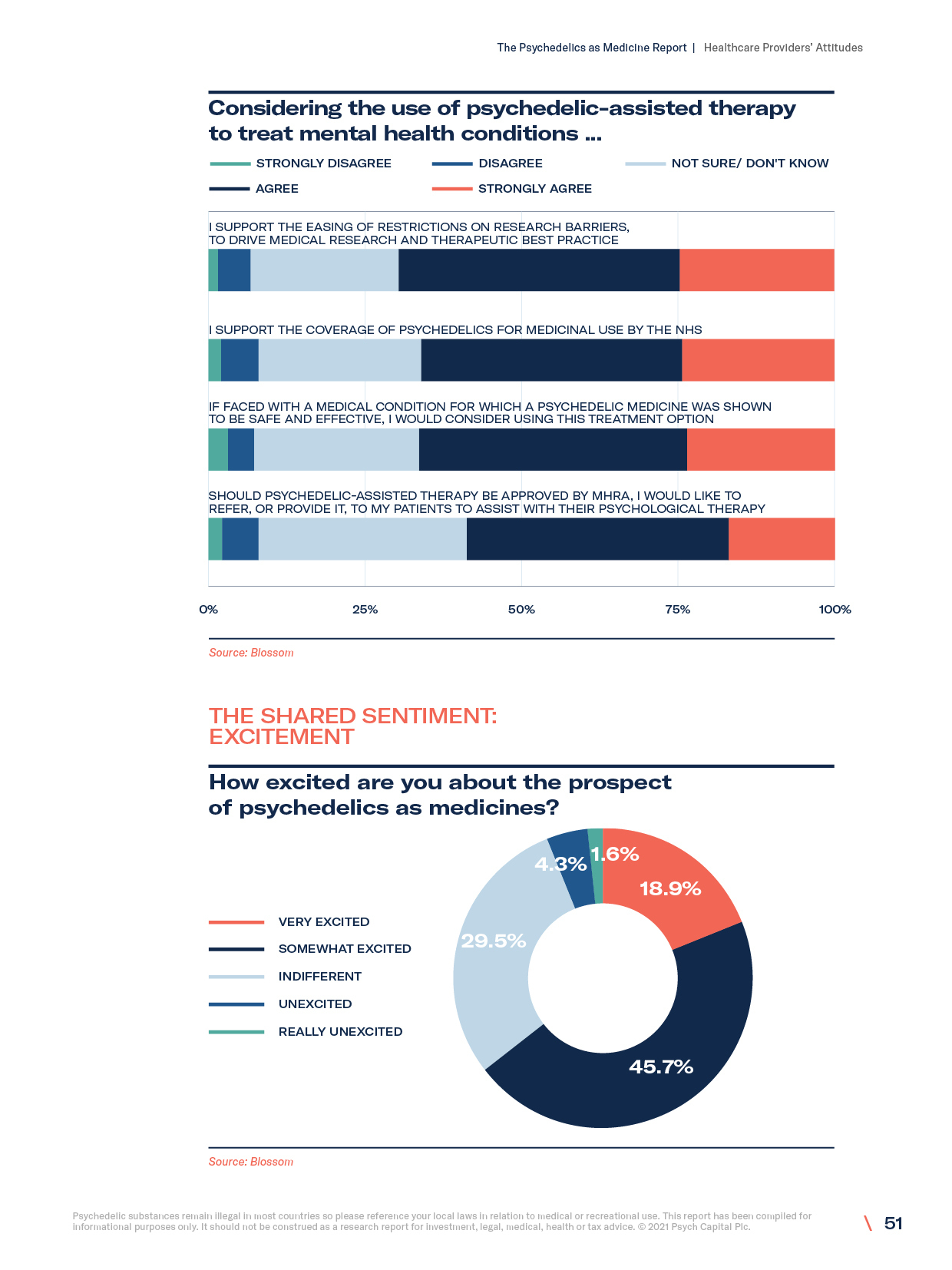
- Healthcare workers’ attitudes towards the medical benefits of psychedelics
- Influence of psychedelic use on practitioner views
Spotlight on Health
- Psychedelics coming out the closet
- Novel ways of treating persistant problems
- Total number of clinical trials
- Key conditions
- Percentage of mental health and substance use disorders
- Facts about mental health conditions
- Psychedelic history and future
- Why Psychedelics are different
Key Psychedelic Deep Dives
- Psilocybin
- LSD
- MDMA
- Ketamine
- Ibogaine
- Ayahuasca
Other Psychedelics of Note
- Salvia Divinorum
- Mescaline
- DMT
- 5-MeO-DMT
- Kratom
- 2C-X
- Nitrous Oxide
Psychedelics per Country
- Australia
- Brazil
- Canada
- China
- Denmark
- Germany
- Ireland
- Netherlands
- Spain
- Switzerland
- United Kingdom
- United States
Expert Contributors
- Florian Brand – Co-founder and CEO, atai Life Sciences
- George Goldsmith – Co-founder and CEO, COMPASS Pathways
- Cosmo Feilding Mellen – CEO, Beckley Psytech
- Dr Roger McIntyre – CEO, Braxia Scientific
- Dr Reid Robison – Chief Medical Officer and Director, Novamind